What is a safe CO2 level in our breathing air?
A lot of literature discusses the quality of breathing gases, contaminants we should be concerned about, how to analyze them, and their toxic effects. There are many standards and guidelines for safe limits, including the results of various occupational health and safety studies on the effects of extended exposure.
For diving, the situation is different. Many compressed-gas safety standards are based only on breathing at or around sea level (1 atmosphere absolute, or ATA). Commercial and navy diving experts have based their limits on contaminant exposure for extended times at depth. So what are safe limits for recreational diving with much shorter exposures than navy divers? Particularly, what is a safe limit for carbon dioxide (CO2), the most abundant potential contaminant?

There are significant discrepancies in international publications, which leads to a question: If the amount of CO2 in compressed air allowed for recreational diving in the U.S. is double the European recommendation, does that mean the U.S. Navy or Compressed Gas Association (CGA)1 standards do not apply to recreational diving? Almost all divers have some knowledge about how CO2 affects them. A safety-conscious diver should also know how to review a filling station’s air-quality analysis certificate with some idea of what to look for and be able to discuss concerns about whether standards are being met or if there is surrounding environmental pollution.
Many studies exist about the physiological effects of elevated CO2 in occupational situations. The results published in health and safety standards and codes of practice throughout the world2 focus on an eight-hour working day. Due to much shorter exposures, recreational divers could, in theory, easily manage significantly higher levels.
Le NOAA Diving Manual (4th edition) describes the relationship between concentrations, exposure limits and symptoms in terms of zones. Table 1 (based on Figure 1) provides a brief correlation between these zones and the units of measure used. The last column indicates the level of CO2 in a scuba cylinder that would produce the corresponding maximum surface equivalent value (SEV). A cylinder containing 1,950 parts per million (ppm)3 of CO2, for example, would achieve an SEV of 15,000 ppm (the upper limit for Zone 1) at a depth of 220 feet of seawater (fsw), 67 meters of seawater (msw), or 7.7 ATA.
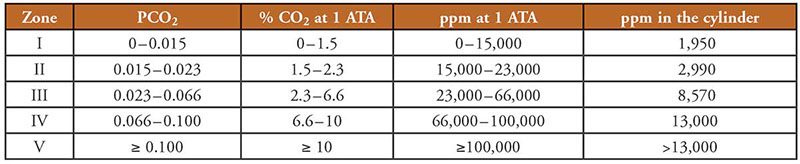
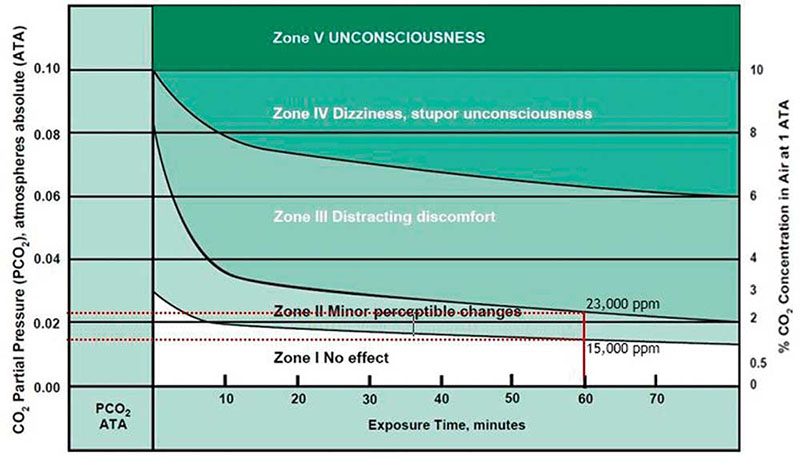
Although these are not trivial values, they have no real meaning to us unless they are associated with the effect on a diver during a typical dive. Figure 1 depicts the effects of a 60-minute dive based on the SEV levels of CO2 contained in a charged scuba cylinder.
Figure 1 illustrates the resulting physiological conditions by zone based on U.S. Navy research, but they do not correlate well with published data from other sources, perhaps because the U.S. Navy expects their divers to be at a much higher level of fitness than the average occupational worker or recreational diver. The value of Figure 1 is in the trend of resulting conditions with the exposure time.
Before deciding on realistic, safe and achievable levels, we must remember that reactions to elevated levels of CO2 vary depending on the individual. The amount of physical activity during a dive has a direct relationship with CO2 production in the tissues and is independent of the levels in the inhaled compressed gas.
Limits should be based on what is safe for a recreational diver who is certified as fit to dive by a qualified physician, assuming relatively strenuous dive activities. The assessment of fitness to dive and what is considered strenuous can be broad and subjective. We can review the research into acceptable CO2 levels under three conditions: at pressure (submarine and navy dive medicine), in an eight-hour working environment (occupational health and safety), and during long-term continuous exposure (NASA space capsule environments). The results indicate that divers can tolerate similar levels to what these conditions cause in most cases.
Maximum depth and bottom time are also important criteria to consider when determining maximum CO2 exposure levels. The deeper we dive, the shorter the bottom time, which the decompression time and the gas used to decompress will determine. A dive to 220 fsw will usually not exceed 15 minutes at this depth unless a longer decompression obligation is accepted. A shallower dive to 60 fsw with a 50-minute bottom time extends exposure but also allows a higher CO2 level to remain within the safe SEV.

CO2 complicates safe exposure because it accumulates in our tissues over time (see Figure 1). The longer the dive, the more CO2 we accumulate and the closer we move toward the next higher zone. To remain in Zone 1 after 60 minutes and avoid symptoms, the CO2 SEV should be no more than 15,000 ppm. To keep within Zone 2, where you may experience minor symptoms, the CO2 SEV should be no more than 23,000 ppm. A CO2 limit would have to apply to all dives, not just those when specific planning considers exposure and time. Limits are there to keep people safe under all expected conditions.
If we transpose the values to cylinder concentration for the deepest exposure (220 fsw), we know that to avoid exceeding Zone 2, the CO2 level in the cylinder should not exceed 2,990 ppm (which equates to an SEV of 23,000 ppm). The maximum value to avoid exceeding Zone 1 would be 1,950 ppm (which equates to an SEV of 15,000 ppm). Based on maximum expected depths (regardless of the breathing gas) and bottom times determined by realistic recreational decompression obligations, these limits are extremely conservative.
Building on these considerations, we can conclude that for regular breathing air during conservative open-water and open-circuit diving, a level of 2,000 ppm in a filled scuba cylinder is considered safe and acceptable. This value encompasses the levels of CO2 currently in the air and local CO2 production, such as from a restaurant or passing vehicles. It is also important to note that sources of carbon monoxide (Sécurité en matière de monoxyde de carbone) and CO2 are not significantly different.
Dive shops should continue to regularly collect and analyze air samples — preferably quarterly but at least annually. CO2 monitors are not cheap, which means that many dive centers and most recreational scuba divers typically would not own an analyzer. Based on only periodic air sampling, a cautious compressed-gas filling facility should trace the source of excess CO2 production. A CO2 value above 400 ppm indicates a level above the standard ambient level. The current CGA limit of 1,000 ppm, even though it is 50 percent higher than the European limit, will most likely not cause adverse effects at depth. This limit is conservative and safe.
Notes
- The current applicable reference in the U.S. is CGA G-7.1-2018 with the commonly referred to Grade E (scuba) air specification of = 1,000 ppmv.
- Several acronyms are found in occupational health and safety standards and guidelines, some indicating instantaneous levels and others the accumulated exposure during an eight-hour working day throughout the life of a worker. The threshold limit value (TLV) describes the upper exposure limit for work in hazardous environments. The permissible exposure limit (PEL) is the legal limit allowed in specific regions or countries. The eight-hour derivative of PEL is the time weighted average (TWA). The recommended exposure limit (REL) is simply a recommended guideline but is intended to supersede the PEL once adopted.
- The term ppm (parts per million) in this article is relative to volume: that is, parts per million by volume (ppmv).
- This figure is a partial replica of Figure 3.11 in the NOAA Diving Manual (4th ed.), originally extracted from the U.S. Navy Diving-Gas Manual (1971). The original data is attributed to Dr. C.J. Lambertsen in the 1971 paper “Carbon Dioxide Tolerance and Toxicity” (Environmental Biomedical Stress Data Center, Institute for Environmental Medicine, University of Pennsylvania Medical Center, Philadelphia; IFEM Report No. 2-71).
- The author correlated the values in this table from data published by the National Oceanic and Atmospheric Administration (NOAA), the National Institute for Occupational Safety and Health (NIOSH), the Occupational Safety and Health Administration (OSHA), the Environmental Protection Agency (EPA) and the National Research Council (NRC). The effects and conditions vary significantly among divers.
- The NRC recommended Emergency Exposure Guidance Level (EEGL) for a one-hour exposure is 25,000 ppm (National Research Council. Emergency and Continuous Exposure Guidance Levels for Selected Submarine Contaminants: Volume 1. Washington, DC: The National Academies Press, 2007. doi:10.17226/11170. Chapter 3 contains a summary of relevant epidemiologic and toxicologic studies on CO2. The wide range of studies reviewed shows significant variations in conditions based on specific exposure levels; the EEGL is very conservative, accounting for the level at which dangerous conditions may present.
© Alert Diver — Q2 2020

